Synaptic remodeling is evolutionarily conserved
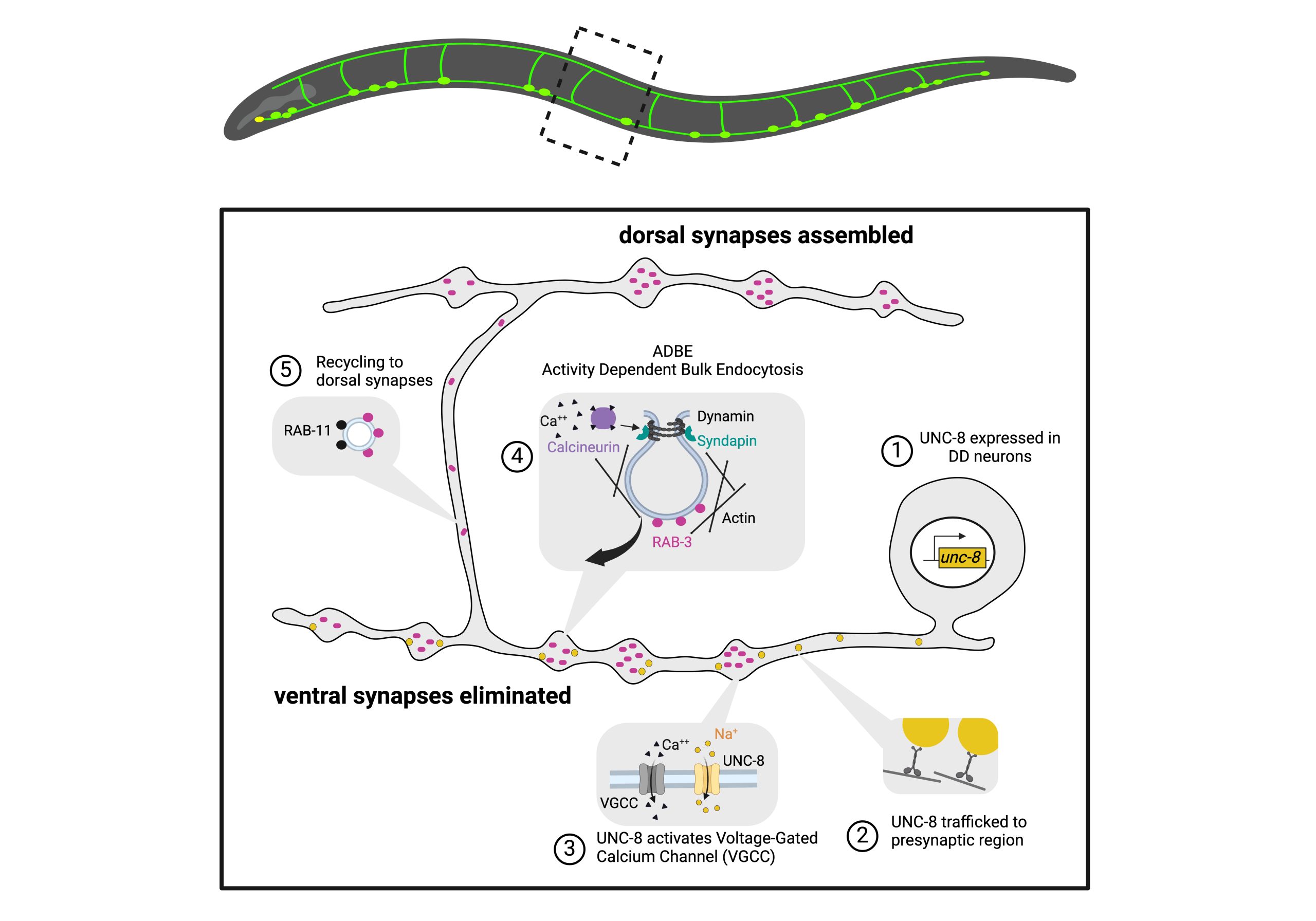
Neurons adopt polarized morphologies to direct the flow of information in the nervous system. The assembly of specialized presynaptic and postsynaptic regions is necessary for the transmission of signals from one neuron to the next. These domains may be remodeled during development or in learning and memory mechanisms. The molecular pathways that govern remodeling are incompletely understood but the conservation of this phenomenon among diverse species suggests that studies in simple model organisms could reveal fundamental elements of synaptic remodeling. The GABAergic motor neurons in the nematode C. elegans display a striking example of developmentally regulated synaptic remodeling. Dorsal D (DD) motor neurons initially innervate ventral muscles but switch polarity to synapse with dorsal muscle during the first larval (L1) stage (White et al., 1978). Ventral D (VD) motor neurons are generated during the L1 stage and adopt the ventral polarity previously assumed by DD motor neurons. Thus, the polarity reversal of DD motor neurons insures that both dorsal (DD) and ventral (VD) muscles receive GABAergic inputs.
We are exploiting this system to uncover genetic factors that govern motor neuron remodeling. These results could lead to significant advances in our understanding of synaptic plasticity and thereby provide a foundation for developing therapeutic approaches for human diseases that disrupt synaptic assembly.
UNC-55 functions as a transcriptional switch to prevent synaptic remodeling
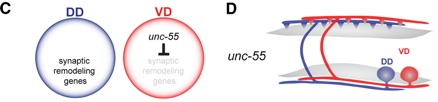
VD motor neurons are prevented from remodeling by the nuclear hormone receptor UNC-55, the C. elegans homologue of COUP-TFII transcription factors (Walthall and Plunkett, 1995,Shan et al, 2005).
In unc-55 mutants, VDs adopt the dorsal axonal polarity normally reserved for mature DDs. Thus, UNC-55 functions as a binary switch in VD motor neurons to block a synaptic remodeling program that results in dorsally directed axonal output. Because UNC-55 is likely to function as a transcriptional repressor, this mechanism is predicted to require UNC-55-dependent repression of synaptic remodeling genes in VD motor neurons.
Expression profiling reveals candidate synaptic remodeling genes
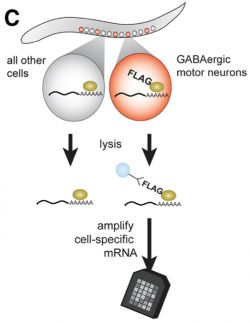
We devised a cell-specific microarray profiling strategy to identify UNC-55 target genes (Petersen et al, 2011). Our approach is based on the assumption that UNC-55-regulated transcripts should be up-regulated (i.e., de-repressed) in unc-55 mutant VD motor neurons. To identify these UNC-55-regulated transcripts, we used the mRNA tagging strategy (Roy et al., 2002; Von Stetina et al., 2007; Watson, et. al., 2008) to compare microarray profiles of wild-type vs unc-55 mutant GABAergic motor neurons. Because UNC-55 is predicted to function as a repressor, we examined genes that were significantly enriched in unc-55 GABA motor neurons.
A transcriptional program remodels GABAergic synapses.
We used the synaptic marker, SNB-1::GFP to mark the axonal domains of DD and VD GABAerigic motor neurons (Hallam and Jin, 1998). In unc-55 mutant adults, SNB-1::GFP puncta are largely limited to the dorsal nerve cord due to ectopic remodeling of VD motor neurons (Fig 1). We reasoned that the absence of ventral SNB-1::GFP puncta should depend on UNC-55 targets and therefore that RNAi of these genes should “suppress” this effect (i.e., restore SNB-1::GFP puncta to the ventral side). RNAi knockdown of candidate UNC-55-regulated transcripts identified in the microarray experiment revealed 50 genes that at least partially suppressed the Unc-55 remodeling defect (p<0.01). Most striking among these candidates is the homeobox transcription factor IRX-1/Iroquois; RNAi knockdown or IRX-1/Iroquois, suppresses both the loss of ventral SNB-1::GFP puncta and the Unc-55 backward movement defect.
Other suppressors of the Unc-55 synaptic remodeling phenotype identified by the RNAi screen include specific ion channels (see below), cytoskeletal components, and cell-cell signaling molecules. The wide range of potential functions encoded by UNC-55-regulated genes is indicative of a complex synaptic remodeling pathway. Ongoing experiments are designed to confirm that these genes function in the unc-55 pathway and to define their cellular roles in the mechanism of synaptic remodeling (Petersen et al, 2011). For a recent review of synaptic remodeling seeCuentas-Condori and Miller, 2020.
The DEG/ENaC cation channel protein UNC-8 promotes activity-dependent synapse removal in remodeling GABAergic neurons.
In additional papers, we confirmed that the UNC-55 regulated target, the DEG/ENaC cation channel protein, UNC-8, promotes the removal of ventral presynaptic domains in remodeling GABAergic neurons (Miller-Fleming, Petersen et al,, 2016;Miller-Fleming, Cuentas-Condori et al., 2021;Cuentas-Condori et al., 2023)