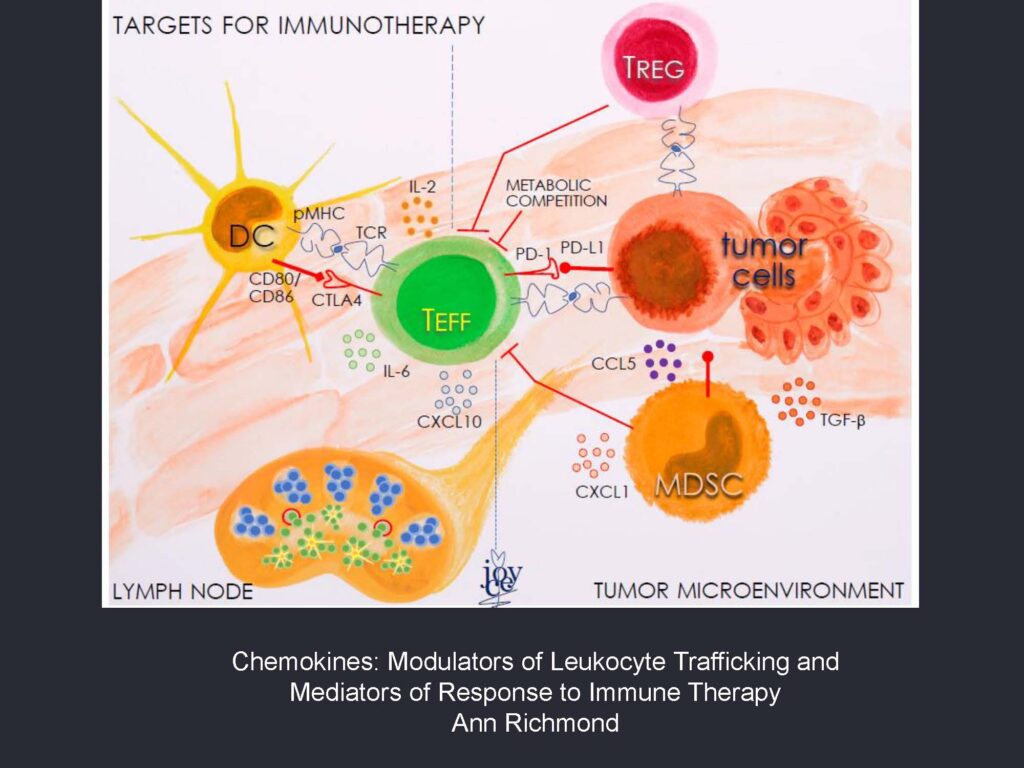
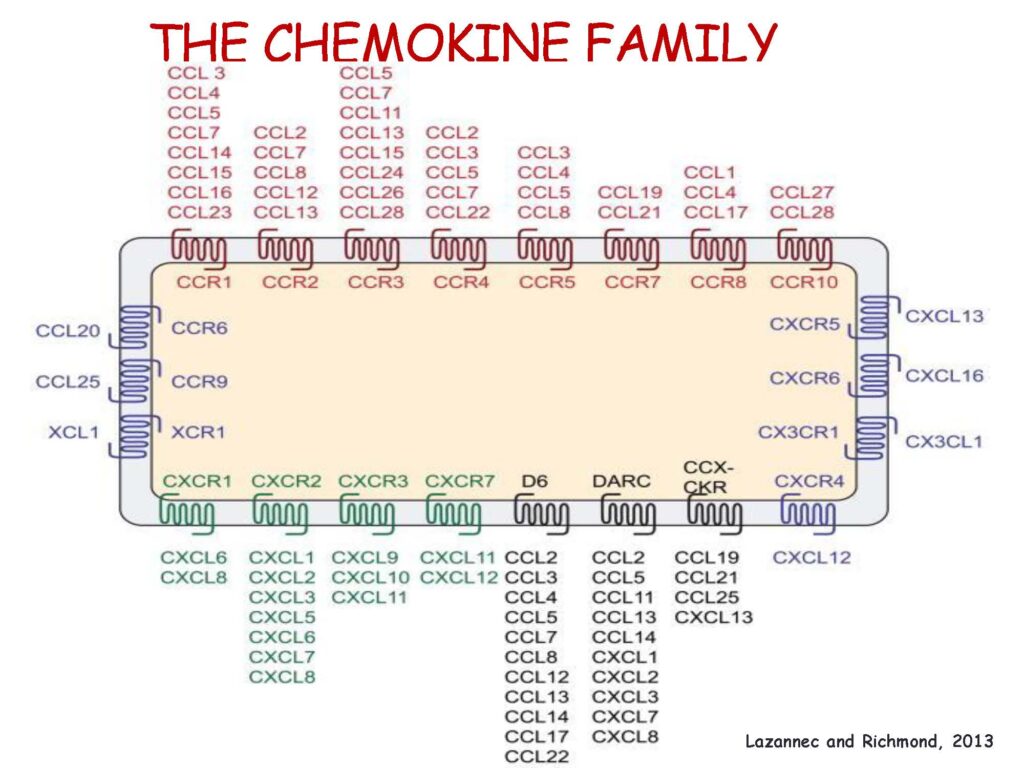
Research

Caroline A. Nebhan MD, PhD and her colleagues published their research in Nature Reviews Clinical Oncology on the implications of long-term toxicity in immune checkpoint inhibitors. Their work found the development of immune-checkpoint inhibitors (ICIs) has heralded a new era in cancer treatment, enabling the possibility of long-term survival in patients with metastatic disease, and providing new therapeutic indications in earlier-stage settings. As such, characterizing the long-term implications of receiving ICIs has grown in importance. An abundance of evidence exists describing the acute clinical toxicities of these agents, although chronic effects have not been as well catalogued. Nonetheless, emerging evidence indicates that persistent toxicities might be more common than initially suggested. While generally low-grade, these chronic sequelae can affect the endocrine, rheumatological, pulmonary, neurological and other organ systems. Fatal toxicities also comprise a diverse set of clinical manifestations and can occur in 0.4–1.2% of patients. This risk is a particularly relevant consideration in light of the possibility of long-term survival. Finally, the effects of immune-checkpoint blockade on a diverse range of immune processes, including atherosclerosis, heart failure, neuroinflammation, obesity and hypertension, have not been characterized but remain an important area of research with potential relevance to cancer survivors. In this Review, we describe the current evidence for chronic immune toxicities and the long-term implications of these effects for patients receiving ICIs. Read more here
 Kennady Bullock, BS (Richmond Lab)
Kennady Bullock, BS (Richmond Lab)
Kennady’s first author paper was published in Cancers in December of 2021. This review explores the immune suppressive role of myeloid-derived suppressor cells (MDSCs). MDSC’s are a heterogeneous population of cells derived from immature myeloid cells. Influx of these cells into the tumor microenvironment or pre-metastatic niche is often associated with poor response to cancer therapy, including immunotherapy, in various tumor types. The C-X-C chemokine receptor 2 (CXCR2) signaling axis plays a key role in the migration of immunosuppressive MDSCs into the tumor microenvironment (TME) and the pre-metastatic niche. MDSCs impede the efficacy of immunotherapy through a variety of mechanisms. Efforts to target MDSCs by blocking CXCR2 is an active area of research for improving existing and novel immunotherapy strategies. As immunotherapies gain approval for a wider array of clinical indications, it will become even more important to understand the efficacy of inhibition of CXCR2 in combating immunotherapy resistance at different stages of tumor progression. Please enjoy the complete article here.
 Kensey Bergdorf Smith, PhD published her first author paper in the April 2022 edition of Cancers (Base). Her research studies the Anaplastic thyroid carcinoma (ATC) is the most aggressive endocrine neoplasm, with a median survival of just four to six months post-diagnosis. Even with surgical and chemotherapeutic interventions, the five-year survival rate is less than 5%. Although combination dabrafenib/trametinib therapy was recently approved for treatment of the ~25% of ATCs harboring BRAFV600E mutations, there are no approved, effective treatments for BRAF-wildtype disease. Herein, we perform a screen of 1525 drugs and evaluate therapeutic candidates using monolayer cell lines and four corresponding spheroid models of anaplastic thyroid carcinoma. We utilize three-dimensional culture methods, as they have been shown to more accurately recapitulate tumor responses in vivo. These three-dimensional cultures include four distinct ATC spheroid lines representing unique morphology and mutational drivers to provide drug prioritization that will be more readily translatable to the clinic. Using this screen, we identify three exceptionally potent compounds (bortezomib, cabazitaxel, and YM155) that have established safety profiles and could potentially be moved into clinical trial for the treatment of anaplastic thyroid carcinoma, a disease with few treatment options.
Kensey Bergdorf Smith, PhD published her first author paper in the April 2022 edition of Cancers (Base). Her research studies the Anaplastic thyroid carcinoma (ATC) is the most aggressive endocrine neoplasm, with a median survival of just four to six months post-diagnosis. Even with surgical and chemotherapeutic interventions, the five-year survival rate is less than 5%. Although combination dabrafenib/trametinib therapy was recently approved for treatment of the ~25% of ATCs harboring BRAFV600E mutations, there are no approved, effective treatments for BRAF-wildtype disease. Herein, we perform a screen of 1525 drugs and evaluate therapeutic candidates using monolayer cell lines and four corresponding spheroid models of anaplastic thyroid carcinoma. We utilize three-dimensional culture methods, as they have been shown to more accurately recapitulate tumor responses in vivo. These three-dimensional cultures include four distinct ATC spheroid lines representing unique morphology and mutational drivers to provide drug prioritization that will be more readily translatable to the clinic. Using this screen, we identify three exceptionally potent compounds (bortezomib, cabazitaxel, and YM155) that have established safety profiles and could potentially be moved into clinical trial for the treatment of anaplastic thyroid carcinoma, a disease with few treatment options.

“Proximity of immune and tumor cells underlies response to BRAF/MEK-targeted therapies in metastatic melanoma patients” Feb. 24, 2022. by Sarah E. Glass
Although targeted therapies have been developed for melanoma patients, tumor progression eventually results. Understanding characteristics of melanoma associated with treatment resistance is important for prolonging therapy effectiveness.
In a retrospective study of patients treated with BRAF and/or MEK inhibitors, Chi Yan, PhD, Ann Richmond, PhD, and colleagues discovered an association between tumor progression and an increase in melanoma cells bearing SOX10, a protein required for melanoma tumor growth.
Reporting in the journal npj Precision Oncology, the researchers found a close physical association of SOX10+ melanoma cells with CD8+ T cells correlated with shorter progression-free survival of patients treated with BRAF/MEK inhibitors.
SOX10 had independent and added prognostic value for late- and early-stage melanoma, respectively: patients with high SOX10/low CD8 tumors had a worse overall survival than those with low SOX10/high CD8 tumors.
While further research is needed, targeting the melanoma – T-cell interaction, particularly through exclusion of SOX10+ melanoma cells or reversion of T-cell suppression, potentially could prevent acquired resistance to BRAF/MEK-targeted therapy.
The research was supported in part by a Merit Award and Senior Research Career Scientist Award from the U.S. Department of Veterans Affairs, and National Institutes of Health grant CA116021.
Ann Richmond, PhD and Chi Yan, PhD recently published their research in Molecular Cancer, “Hiding in the dark: pan-cancer characterization of expression and clinical relevance of CD40 to immune checkpoint blockade therapy”.
Their work highlights that CD40 expression correlates with the type I anti-tumor response and better survival. Pan-cancer bioinformatics characterization reveals it reduced CD40 expression in 11 cancer types, including RASmut melanoma compared to nevi. Furthermore, RAS mutation correlates with reduced CD40 expression in malignant melanoma, and that CD40 expression is associated with a better response to immune checkpoint blockade therapy in melanoma. This work was funded by grants from the Department of Veterans Affair and the NCI.
“Preclinical study supports rigosertib as booster to immunotherapy treatment for melanoma” Jul. 1, 2021
A team of Vanderbilt researchers have shown in a preclinical study that the investigational drug rigosertib could be a potential booster treatment to elicit response to immunotherapies among melanoma patients.
While immune checkpoint blockade therapies are already an established first-line treatment for metastatic melanoma patients, these immunotherapies are effective for about 52% of patients. The team of researchers, utilizing mouse models and patient-derived xenografts, investigated whether rigosertib would enhance response to these immunotherapies.

Their study, published online June 6 in Molecular Cancer, indicated that rigosertib was well tolerated in mice, inhibited tumor growth by about 50% as a stand-alone treatment and by about 70% when combined with anti-PD1 and anti-CTLA-4 immunotherapies.
The investigators also found that rigosertib, or combination of the BRAF/MEK targeted therapies dabrafenib and trametinib, increased levels of a CD40 protein. High levels of this protein correlate with beneficial T-cell responses and better survival. CD40 expression by melanoma cells is also associated with therapeutic responses to immune checkpoint blockade immunotherapies.
The study reveals a novel role of RAS/RAF/P13K inhibition for inducing immunogenic cell death by inducing CD40 expression on melanoma cells as well as inducing an anti-tumor immune response. More importantly, it provides preclinical evidence for the combination of rigosertib and immune checkpoint blockade treatment as potential therapy for patients with metastatic melanoma. This is especially important since melanoma patients who do not have BRAF mutation but exhibit NRAS mutation, or have neither BRAF nor NRAS mutation, can be treated with rigosertib.
“We are very encouraged by these findings and expect a new clinical trial to launch soon combining rigosertib with anti-PD-1 for melanoma patients who are resistant to anti-PD-1 therapy alone. We are working with Drs. Doug Johnson and Caroline Nebhan to launch this trial”, said senior author Ann Richmond, PhD, professor of Pharmacology and Ingram Professor of Cancer Research.
The study’s lead author is Chi Yan, PhD, who designed the research approach with Richmond, and performed the experiments with Nabil Saleh and Jinming Yang, PhD ,with assistance from Caroline Nebhan, MD, PhD, Anna Vilgelm, MD, PhD, E. Premkumar Reddy, PhD, Joseph Roland, PhD, Douglas Johnson, MD, MSCI, Sheau-Chiann Chen, PhD, Rebecca Shattuck-Brandt, PhD, and Gregory Ayers, MS.
Article written by Tom Wilemon
Study reveals new strategy for reducing tumor growth, metastasis.
Dec. 17, 2020
by Leigh MacMillan
A team of Vanderbilt investigators has discovered that blocking a certain signaling pathway boosts antitumor immunity and reduces tumor growth and metastasis in models of breast cancer and melanoma.
The findings, reported in the journal Cancer Immunology Research, add mechanistic insight and support for the therapeutic potential of drugs that are currently being tested in clinical trials for various types of cancer.
The studies focus on the complex interplay of signaling factors and immune system cells in the tumor microenvironment. To grow and survive, a tumor must evade the body’s immune response against it. One way tumors do this is by secreting factors that recruit a set of cells called myeloid-derived suppressor cells (MDSCs), which originate in the bone marrow and can suppress the T cell-mediated immune response.
Ann Richmond, PhD, Ingram Professor of Cancer Research and professor of Pharmacology, and her team have long studied tumor-secreted chemokine factors and their receptors on MDSCs, in particular the receptor CXCR2. CXCR2 is a key chemokine receptor involved in the movement of MDSCs into developing tumors and the pre-metastatic niche.
“We had the hypothesis that if we could target CXCR2 in myeloid cells, we might ablate the trafficking of MDSCs into the tumor microenvironment and thereby enhance the immune response to a developing tumor,” Richmond said.
The researchers, led by Jinming Yang, PhD, staff scientist, and Chi Yan, PhD, research instructor in Pharmacology, did just that.
They developed a mouse model with a targeted deletion of the CXCR2 gene in myeloid cells and studied the development of tumors from implanted breast cancer or melanoma cells. They found reduced tumor growth and metastasis for both types of tumors in mice missing CXCR2 signaling in myeloid cells.
An in-depth analysis of immune cells and chemokine factors in the tumors revealed surprises, Richmond said. In addition to a reduction in MDSCs in the tumors, the researchers found increased numbers of B cells (of the B1b type) and CD8+ T cells, which kill tumor cells, along with increased levels of the chemokine CXCL11.
“This is a paradigm shift — the idea that by simply blocking CXCR2 expression by myeloid cells, there is an influx of B cells making CXCL11, which then recruits T cells into the tumor microenvironment,” Richmond said. “Moreover, since the loss of CXCR2 expression in myeloid cells led to depletion of MDSCs, there is also reduction in MDSC suppression of the CD8+ T cell antitumor activity.
The final result is stronger antitumor immunity that suppresses tumor growth and metastasis.”
The researchers demonstrated that the effect was dependent on both the B cells and the CD8+ T cells and that when either of these cell populations were depleted from mice missing CXCR2 expression in myeloid cells, the inhibition of tumor growth was markedly reduced.
Importantly, Richmond noted, using systemic delivery of a drug that blocks the CXCR2 receptor produced the same effects in tumors implanted in control mice.
“The CXCR2 antagonist we used is currently in clinical trials in combination with immune therapy for patients with melanoma, and our studies have now substantiated the intricate mechanism that’s involved,” Richmond said. “Patients don’t all respond to any one therapy, so being able to see how these various immune cells are being modulated with CXCR2 antagonist provides mechanistic insight into how CXCR2 antagonist combined with immune checkpoint inhibitor therapies might work in patients.”
To explore whether the B cell-CXCL11 signaling axis may be important in breast cancer and melanoma prognosis, the researchers queried databases of human molecular cancer data (TCGA and GEO-NCBI). They found positive correlations among B cell markers, CXCL11 and a CD8+ T cell marker in association with increased survival among breast cancer and melanoma patients.
“These data appear to confirm that B cells and CXCL11 are working together to bring in more T cells to enhance tumor killing and survival,” Richmond said.
Moving forward, the researchers are testing combinations of CXCR2 inhibitors, immune therapies and other signaling pathway inhibitors in various cancer models.
Other Vanderbilt investigators contributing to the studies included Anna Vilgelm, Sheau-Chiann Chen, Gregory Ayers and Christopher Johnson. The research was supported by grants from the National Institutes of Health (CA034590, CA243326, CA116021, CA006845, CA233770) and from the U.S. Department of Veterans Affairs.
Investigational agent called KRT-232.
 Recent research from the lab of Ann Richmond tested an investigational agent called KRT-232 to see if mice responded better to it compared to treatment with the standard drugs. They found that the mice fell into 3 groups: (1) those that responded best to the standard drugs but not to KRT-232, (2) those that responded well to the standard drugs AND KRT-232 together, and (3) those that responded to KRT-232 alone, but did not benefit from the other drugs.
Recent research from the lab of Ann Richmond tested an investigational agent called KRT-232 to see if mice responded better to it compared to treatment with the standard drugs. They found that the mice fell into 3 groups: (1) those that responded best to the standard drugs but not to KRT-232, (2) those that responded well to the standard drugs AND KRT-232 together, and (3) those that responded to KRT-232 alone, but did not benefit from the other drugs.
~
This research shows that this new drug, KRT-232, is promising for treating patients that don’t respond to the standard therapy! Even better: the Richmond group found that certain genetic mutations — including one that’s routinely screened already! — could serve as an indicator for which patient will respond to which therapeutic strategy. Vanderbilt Basic Sciences on Instagram: “Current treatments for metastatic melanoma, an aggressive type of skin cancer, work well for about 60% of patients. The American Cancer…”

Immunotherapy and the History of Chemokines